 
It can be shown as an image or a picture. The free-moving electrons hit the fluorescent tube on the other side of the glass tube and thus cause momentary flashes which can be electrically monitored. He was a German physicist who developed the device using a glass envelope or a glass tube with one end containing an electron gun that traverses within the tube. The cathode ray tube, which is also known as CRT, is a device that was invented in the year 1897 by Karl Ferdinand Braun. NCERT Solutions for Class 10 Social Science.NCERT Solutions for Class 9 Social Science.NCERT Solutions for Class 8 Social Science.NCERT Solutions for Class 7 Social Science.NCERT Solutions for Class 6 Social Science.JEE Advanced Previous Year Question Papers.This photograph is in the public domain.Agra,ahmedabad,ajmer,akola,aligarh,ambala,amravati,amritsar,aurangabad,ayodhya,bangalore,bareilly,bathinda,bhagalpur,bhilai,bhiwani,bhopal,bhubaneswar,bikaner,bilaspur,bokaro,chandigarh,chennai,coimbatore,cuttack,dehradun,delhi ncr,dhanbad,dibrugarh,durgapur,faridabad,ferozpur,gandhinagar,gaya,ghaziabad,goa,gorakhpur,greater noida,gurugram,guwahati,gwalior,haldwani,haridwar,hisar,hyderabad,indore,jabalpur,jaipur,jalandhar,jammu,jamshedpur,jhansi,jodhpur,jorhat,kaithal,kanpur,karimnagar,karnal,kashipur,khammam,kharagpur,kochi,kolhapur,kolkata,kota,kottayam,kozhikode,kurnool,kurukshetra,latur,lucknow,ludhiana,madurai,mangaluru,mathura,meerut,moradabad,mumbai,muzaffarpur,mysore,nagpur,nanded,narnaul,nashik,nellore,noida,palwal,panchkula,panipat,pathankot,patiala,patna,prayagraj,puducherry,pune,raipur,rajahmundry,ranchi,rewa,rewari,rohtak,rudrapur,saharanpur,salem,secunderabad,silchar,siliguri,sirsa,solapur,sri-ganganagar,srinagar,surat,thrissur,tinsukia,tiruchirapalli,tirupati,trivandrum,udaipur,udhampur,ujjain,vadodara,vapi,varanasi,vellore,vijayawada,visakhapatnam,warangal,yamuna-nagar Classroom top menu.PBS atom builder References References for this article 
Measured masses and charges of the three elementary particles are given in the following table.For a neutral atom, number of proton=number of electron.Atomic mass = mass of protons mass of neutron.The gold foil experiment was originally conducted by Hans Geiger (left) and Ernest Marsden (right) under the supervision of Ernest Rutherford at the University of Manchester. James Chadwick (on the right), a student of Rutherford won Nobel Prize in Physics in 1935 for discovery of neutron. Ernest Rutherford (left) was awarded Nobel Prize in Chemistry in 1908 for his work in radioactivity. Here the symbol  \(_Z^XA\) is used where Z = atomic number and X = atomic mass of the element A. Figure 7. Thomson measured the charge-by-mass-ratio (e/m) of cathode ray particle using deflection in both electric and magnetic field. 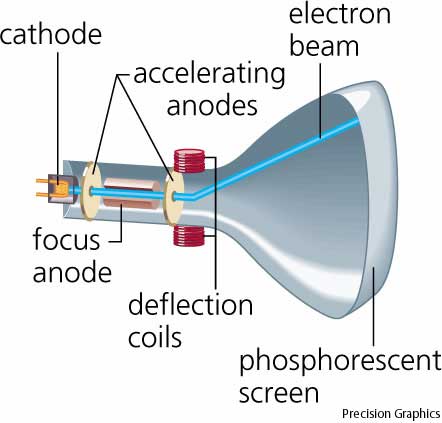
Deflection of cathode rays towards positively charged plates Hence cathode ray consists of negatively charged particles. Applying electric field in the path of cathode ray deflects the ray towards positively charged plate. They are independent of the material composition of the cathode.ģ. The schematic of a cathode ray tube is given in figure 2. This ray is called cathode ray and the whole construction is called cathode ray tube. He detected that a stream of particle (ray) was coming out from the negatively charged electrode (cathode) to positively charged electrode (anode). Then he applied a high electrical voltage between two electrodes at either end of the tube. much of the air was pumped out of the tube. Thomson constructed a glass tube which was partially evacuated i.e. Thomson also won the Nobel Prize in 1937 for proving the wavelike properties of electron. Thomson won Nobel Prize in 1906 for discovering the elementary particle electron. Thomson in 1897 when he was studying the properties of cathode ray. Discovery of Electron Who discovered the electron?Įlectron was discovered by J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
